 Greenbarn Potter's Supply Ltd.
Greenbarn Potter's Supply Ltd.

Normal Hours of Operation Open Mo-Fr 8:30-5PM, closed Sat, Sun and long weekends. Next closure for stat holiday is for Easter, closed on April 3 and April 6, and opening with regular hours on April 7, 2026. Information Plainsman Products ClaysLow Temperature ClaysMedium Temperature Clays High Temperature Clays Porcelains Other Clays Casting Slips MaterialsDry MaterialsStains Encapsulated Stains Liquids GlazesSpectrum Low Stone GlazesLaguna Dry Low Fire Glazes Spectrum Opaque Gloss Low Fire Glazes Spectrum Semi-Transparent Low Fire Glazes Spectrum Satin Matte Low Fire Glazes Spectrum Crackle Glazes Spectrum Metallic Glazes Spectrum Raku Glazes Plainsman Dry Glazes Potter's Choice Cone 5/6 Glazes Celadon Cone 5/6 Glazes Moroccan Sand Glazes Spectrum Hi Fire Cone 6 Glazes Spectrum Shino Glazes Cone 6 Spectrum Celadon Glazes Cone 6 Liquid Brights UnderglazesSpectrum 500 UnderglazesUnderglaze Tools Amaco Velvet Underglazes EnamellingEnamelling SuppliesEquipmentKilnsElectric Pottery KilnsElectric Glass Kilns Kiln Furniture Cones Elements Kiln Parts, Accessories Exhaust Systems Refractories Potter's Wheels Slab Rollers Hand Extruders Pugmills Scales Banding Wheels Air Brushes ToolsBrushesThrowing Tools Trimming, Turning, Cutting Tools Wood/Bamboo Tools Wire and Wood Tools Rollers/Stamps Decorating Tools Glazing Tools Ribs & Scrapers Ribbon/Wire Tools Rasps Knives, Needle Tools, Cutters Sculpture Tools Tool Kits Unclassified AccessoriesMiscellaneous AccesoriesCorks/Stoppers Cork Pads Oil Lamp Accessories Dispenser Pumps Teapot Handles Bisque Tiles | Visit Our Full Catalog & Store Western Canada's largest distributor of pottery materials and supplies. Clays, raw materials, tools, wheels, kilns, slabrollers, books & much more. Our continuing goal is to supply artists, potters and crafts people with great quality products, knowledge and customer service. Our staff is familiar with all the items we stock and can help you through the selection and ordering process. We will also see that your order is shipped according to your directions, or put together for pick up at our retail store in Surrey, BC. Canadian Electrical Standards - A PSA on CSA Greenbarn would like to take a brief moment to highlight CSA. CSA, or the Canadian Standards Association, is a non-profit organization that develops and publishes standards for a wide range of products and services to improve safety, health, and sustainability in Canada and beyond. In the pottery world, CSA approval for electrical products such as pugmills, kilns and wheels helps to protect Canadian artists. Specifically, CSA protects us against physical hazards like electrical shock and fire, but also against financial hazards like fines, penalties, or the possibility of our home insurance's refusal to pay in the case of any damage by fire. Obtaining CSA certification is a mandatory, legal requirement by all retailers within Canada. It demonstrates our collective commitment to high quality products, it provides safety and confidence to consumers, and it also demonstrates to regulatory bodies that our products meet stringent Canadian safety standards. Firstly, we would like you to feel confident that Greenbarn always adheres to the legal requirement that our equipment be CSA inspected and approved. Each piece of equipment will have a sticker which proves if it has been successfully tested. Secondly, while Greenbarn is incredibly appreciative of your support, we would like to arm our community with the knowledge to internet shop safely. We have noticed that not all retailers follow their legal obligation, and we wish to advocate for our customer's safety by recommending that wherever you choose to purchase your electrical products, please know that CSA approved equipment protects you, and if it is unclear, it is your legal right to request proof of this level of safety in Canada. We hope this is helpful for you. Thank you for reading, and for your support! - Greenbarn Enamel Powders and Accessories on SALE at 25% off! Thompson brand copper enameling materials and tools are now on sale at 25% off. These can be great for your school art/metal projects, your mixed media crafts, and for jewelry. Once the enamel powder has been applied to the copper shape, fire it with a kiln, or a torch, to 1450-1500*F to melt the enamel into a glass finish. Use our search box and enter "enamel", or use our website index in the left margin and click on "Enamelling" to see the shapes, tools and enamel pigments.
Technical Tips BlogWhen kilns are not candled long enough
Candling of kilns is the final stage of drying. Driers cannot achieve the temperatures needed to remove all water, so almost all industries rely on early stages of firing to remove it fully. Failures like this are part of the learning curve of every company (because there is always pressure to fire as fast as possible). Context: This is what happens.., Automated porcelain insulator trimming.., Dehydroxylation in kaolin ball.., Pore water removed in.., Candling Friday 27th March 2026 Paint another layer on a fired glaze?Yes. With CMC gum.
The cone 6 mug on the left has the G3933A glaze, applied as a dipping glaze. It turned out poorly - crawling from corners and looking thin and washed out. I made a brushing glaze version of this (which adds 1.5% CMC gum), I keep it around for this very purpose. It has a high specific gravity (unlike commercial ones that have high water contents - they will run and go on too thin if you try this). Because of the gum, it dries hard, there is no shrinkage or cracking. On a second firing, using the C6DHSC schedule again, (mug on the right) the surface is transformed - thicker, more vibrant color. Context: CMC Gum, Six layers 85 Alberta.., Control gel using Veegum.., The degree-of-matteness of this.. Thursday 26th March 2026 2% Copper carbonate in two cone 6 transparents:One does not bubble and orange-peel. Why?
The top base glaze, G2926B, has enough melt fluidity to produce a brilliant functional gloss when used as a transparent. However, for this 2% copper carbonate addition, it has too little melt fluidity and/or too much surface tension to merge, pass and heal the entrained micro-bubbles (generated by the decomposition of the carbonate). Context: Copper Carbonate, Copper Carbonate Basic, Zinc Oxide, ZnO, G3806C, Why this copper glaze.., A light bulb moment.., Flux Thursday 26th March 2026 A Clear Glaze is Bubbled Over DIY Underglazes:The Real Solution Is in the Underglaze Recipe
Potters often encounter the problem shown here. These pieces are fired at cone 6. They are decorated with underglazes made from a mix of porcelain powders and stains. The transparent glaze works over certain colors (e.g. the light blue), but over others, it is full of microbubbles and pinholes. The potter has not had success finding a transparent overglaze that works consistently. As can be seen here, stain types used in underglazes behave differently; they are not just inert powders. Also, stain manufacturers do not mix stains with porcelain to making underglazes. Context: A 2oz jar of.., Here is another reason.., Underglaze, Stain Medium Saturday 21st March 2026 DIY the commercial glaze on mug #1:You must consider five factors to make it work
The mug on the left, #1, is a commercial brushing glaze. It is opaque enough to cover this red-burning clay body. It shows the desired effect. That depends on the fact that opaque glazes stretch thinner on the sharp edges of incised designs. If they have enough melt mobility and are applied right, the effect is amplified. This potter is attempting to mix her own DIY equivalent as a dipping glaze, adding 4% tin oxide to a transparent base glaze in #2 and zircon (a higher percentage) in #3. As you can see, the effect is not working as well, and there are several reasons: Context: A comparative glaze opacity.., Opacity, Opacifier Thursday 26th February 2026 Multilayer crawling
Oil-spot effects depend on being able to layer glazes. Normally, a black underneath and matte white over top. Using dipping glazes is obviously advantageous for this type of piece; the second dip covers the other half and creates the double layer needed. But dipping glazes contain clay in the recipe (almost always kaolin or ball clay), it satisfies multiple needs: It suspends the slurry, hardens the drying glaze, and supplies critical Al2O3 to the chemistry. The upper glaze here is a matte; so it needs extra Al2O3, which means it likely has extra clay. 20% kaolin (non-plastic like EPK, Grolleg, NZ) is about the maximum or the glaze will shrink too much when drying. If extra Gerstley Borate is added, then it will be worse. Drying cracks are the result when the fragile body-bond of the lower one fails to withstand the stresses of being rewetted and tugged upon by the upper layer being applied and drying. When the cracked double-layer begins to melt, it pulls itself into islands, leaving bare body between. That is called "crawling". Context: Glaze Layering, Crawling Tuesday 17th February 2026 Tile stacking in an electric kiln - Fingers crossed!
Small-scale operations everywhere are making tile like this. Most use plastic clay intended for pottery, which introduces more drying shrinkage, complicating drying them flat. Stacking them in the kiln can be a game of chance. Stacked too tightly and they crack (mostly because of quartz inversion). Stacked to loosely and most of the energy goes into heating the shelves and stackers. Using a clay with minimal large quartz particles is the best way to avoid dunting, however that is also a balance since such clays are more difficult to fit glazes to (without crazing). Context: Tile having angular shape.., An unevenly cooled tile.., It possible to make.., A plastic pottery clay.. Tuesday 17th February 2026 Stains are better in black DIY glazesUse 5% stain instead of 15% metal oxides
Consider the hazards and hassles before choosing a black matte or gloss recipe that has high individual or combined percentages of manganese dioxide, cobalt or nickel. Context: Ceramic Stain Toxicity Label.., Two cone 6 black.., Heres evidence that using.., Ceramic Stain, Toxicity Monday 16th February 2026 A plastic pottery clay for rolling ceramic tile:Not a crazy idea when it can do what this can!
This is the dolomite body recipe L4410P (a development version of Plainsman Snow). It is monoporosa tile on steroids; this body has zero percent firing shrinkage at cone 04! Predicting the final size and keeping that size consistent is much easier with such bodies. I have measured its drying shrinkage as 6% (doing our standard SHAB test). The final size needed is 20.5 cm square. Thus, I calculate the cut size to be 20.5 / (100 - 0.06) = 21.8 cm (or 20.5 / 0.94 = 21.8 cm). To keep these flat, we put them between sheets of drywall; the process takes 2-3 days. Since no change in size occurs during firing, this body has another big advantage: Tiles stay flatter during firing (a major problem with tile production). While making wall tile using a plastic pottery body is not something for industry (especially because of the space requirements for drying), for artisans working on a small scale, a body made by mixing super plastic ball clay with dolomite produces amazing working and tactile properties. The bonus is that they work so well at low temperatures, where there are so many glazing options. Context: Monoporosa or Single Fired.., QRCode mounted on Plainsman.., Tile stacking in an.., Tile that is actually.., Ceramic Tile Sunday 15th February 2026 This boron blue effect depends on three things:A dark body, variations in thickness, the right chemistry
This is G2826A3, a transparent amber glaze at cone 6 on white (Plainsman M370), black (Plainsman 3B + 6% Mason 6666 black stain) and red (Plainsman M390) stoneware bodies. When the glaze is thinly applied, it is transparent. But at a tipping-point-thickness, it generates boron-blue that transforms it into a milky white. Glazes that are very glassy but on the edge of structural instability do this. So they are not good for functional ware. Context: A pottery glaze that.., Boron Blue, Glaze thickness Saturday 14th February 2026 I Tested a Found-Clay:Was it suitable for pottery?
Would you like to be able to use your own found-clays, ones native to your area or even your property, in your production? Follow me as we evaluate a mystery clay sample provided by a potter who wants to do exactly this. I will use ordinary tools that any potter either already has or can buy at low cost. We will describe this clay in terms of plastic clay bodies and common ceramic materials that most potters already use. The potter who submitted it has worked enough with the material to suspect it has potential and he wants to know how to best utilize it (e.g. at what temperature, with what glazes, mixed with what, processed in what way). In technical terms what we are doing is called "characterization". Context: Evaluating a clay's suitability.. Thursday 12th February 2026 Tile that is "actually HANDMADE"
This artisan, Dennis Cuku, is the king of DIY tile, making product that he calls "actually HANDMADE". He uses a red-burding terra-cotta-like middle temperature clay body. He also makes glazes in-house and fires using 36 shapes. He mixes 129 glazes and produces about 50,000 ft.² of tile per year. Tile making presents many unique challenges, not the least of which is the need for consistency and predictability of surface character and color. This endeavour is made possible with data, a lot of it. Not just glaze recipes, but many forming, glazing and firing procedures and techniques that must be documented. Context: Potters can learn from.., Making complex ceramic tile.., A plastic pottery clay.. Saturday 7th February 2026 Fine-tuning the thixotropy of a glaze or engobeFor dipping, this is so much better!
Watch this 30-second video to see. Gelled (thixotropic) slurries for dipping are so much better to work with; you'll never go back once you have mastered this DIY technique. While some glazes and engobes gel naturally, especially those with high clay content, these almost always work best when the water content is within a certain range, so fine-tuning like this is still needed. Although not shown here, if over-gelling happens, a drip or two of deflocculant (e.g. Darvan) brings back the fluidity, this is more likely to happen with engobes since they need more gel (for dipping and even more for painting). A side benefit of this: No settling in the bucket. Context: Fine tune the thixotropy.. Wednesday 4th February 2026 Phase separation close-upThe power of modern phone cameras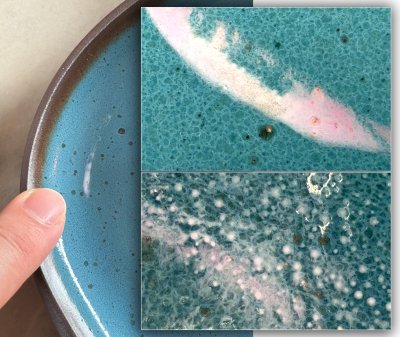
This reduction stoneware glaze is producing white streaks on some pieces (left center). The body is a coarse iron stoneware. A magnification is needed to better explain this. Context: New macro-capable cameras on.., Phase Separation Friday 30th January 2026 Cone 6 iron red with a catcher glaze
This is G3948A (similar to the popular Amaco Ancient Copper product). To get this stunning result, it needs to be applied thickly. Therefore, it runs a lot. But the catcher glaze around the bottom of these mugs has stopped the flow. The catcher is a glossy black, G3914A (but Amaco Obsidian would also likely work). I have learned to put it on with the right height (about 2cm) and right thickness, and then apply wax emulsion to prevent the iron red glaze from sticking during dipping. The inside glaze, G2926B, is one I have tested and developed to fit Plainsman clay bodies as a liner. Context: You can make your.., Industry can never make.., Catch Glaze Thursday 15th January 2026 |
Greenbarn Potter's Supply Ltd., 9548 - 192nd Street, SURREY, BC V4N 3R9
Phone: 604-888-3411, Email: sales@greenbarn.com